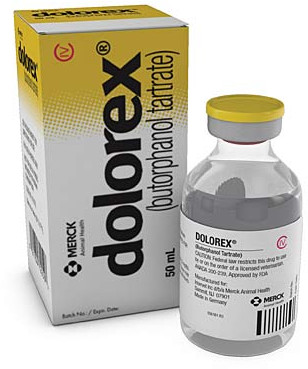
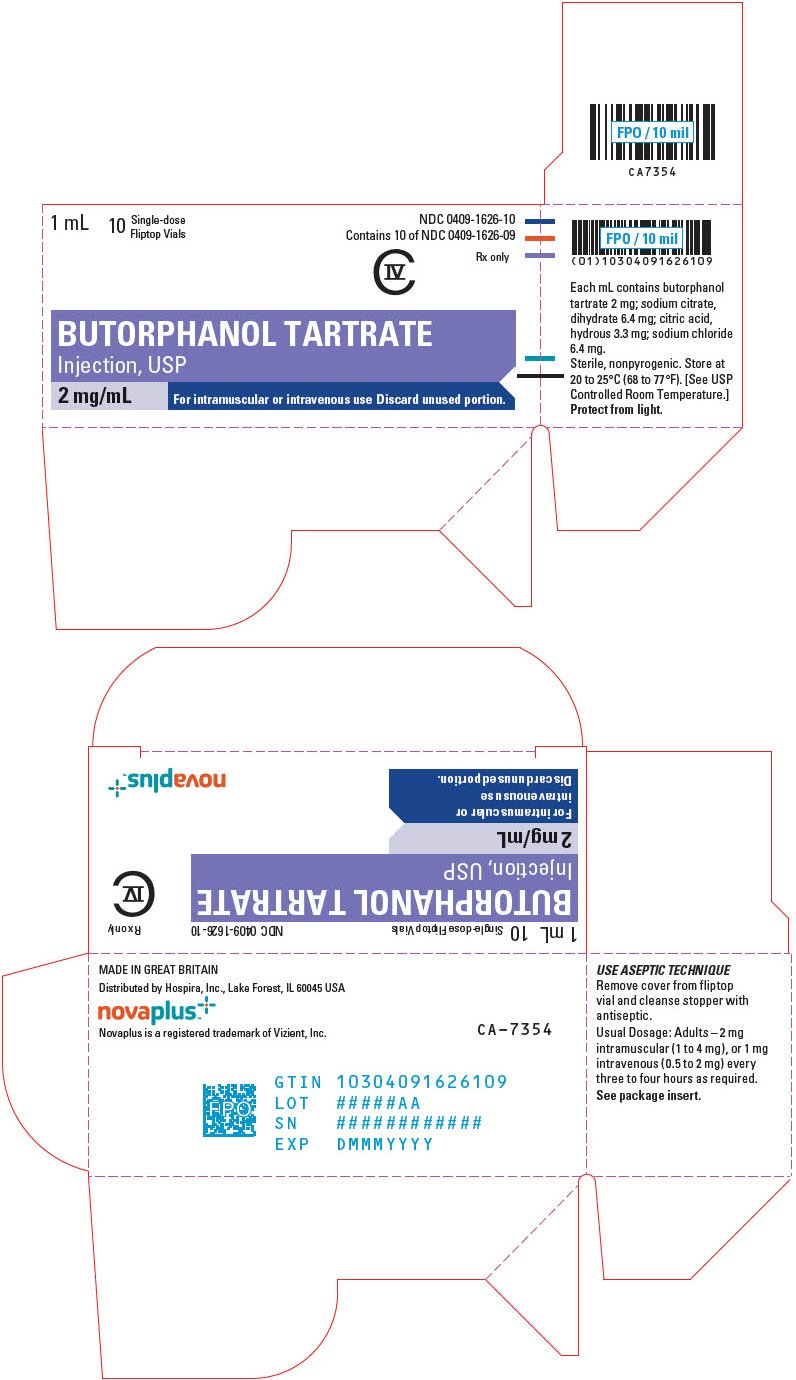
Stadol Nasal Spray Discontinued
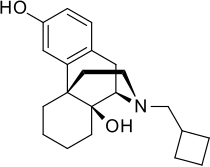
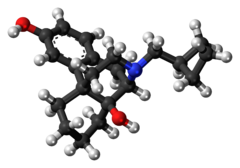
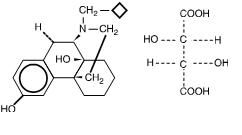
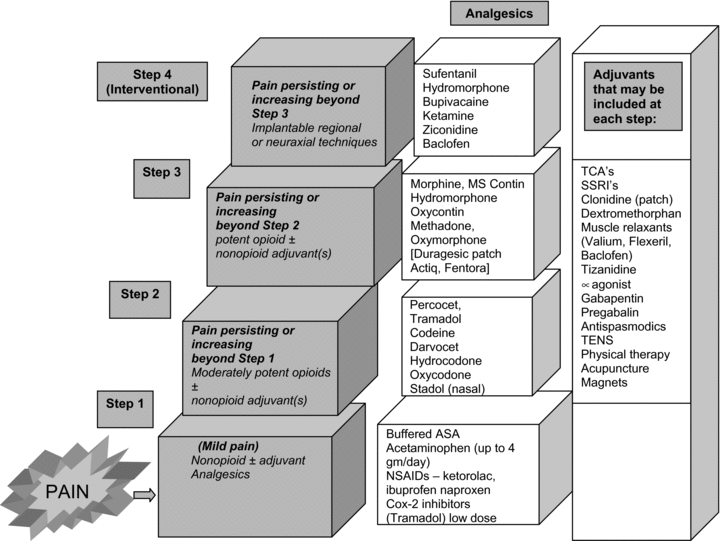
Stadol nasal spray discontinued. Adherence to this dose reduces the incidence of drowsiness and dizziness. It is used to treat moderate to severe pain. Prior Approval is required only in order to EXCEED the standard allowance Stadol Nasal Spray butorphanol NOTE.
1MGSPRAY Federal Register determination that product was not discontinued or withdrawn for safety or efficacy reasons SPRAY METEREDNASAL. BUTORPHANOL is a pain reliever. 5 Lidocaine and 75 Dextrose Injection.
Sleep problems insomnia. Most of these patients abruptly discontinued butorphanol tartrate nasal spray after extended use or high doses. You may report side effects to FDA at 1-800-FDA-1088.
234 Sodium Chloride Injection. Nasal spray for patients that are 18 years of age or older. It is also used to prevent pain before surgery and during child birth.
The Stadol brand name has been discontinued in the US. Butorphanoladministration. 09 Sodium Chloride Small Volume Bags 150 mL Apr 15 2021.
If generic versions of this product have been approved by the FDA there may be generic equivalents available. Find information about which conditions Stadol NS nasal is commonly used to treat. 8496065 PubMed - indexed for MEDLINE Publication Types.
Drowsiness dizziness blurred vision flushing headache nausea vomiting constipation nasal irritationcongestion trouble sleeping dry mouth and. BUTORPHANOL is a pain reliever.
Https Www Researchgate Net Publication 323659139 What S New In Critical Illness And Injury Science The Quest For Effective And Safe Co Induction Agents In Spontaneously Breathing Patients Undergoing General Anesthesia Fulltext 5aa2815ea6fdcc22e2d2f2a2 Whats New In Critical Illness And Injury Science The Quest For Effective And Safe Co Induction Agents In Spontaneously Breathing Patients Undergoing General Anesthesia Pdf
Https Www Researchgate Net Publication 323659139 What S New In Critical Illness And Injury Science The Quest For Effective And Safe Co Induction Agents In Spontaneously Breathing Patients Undergoing General Anesthesia Fulltext 5aa2815ea6fdcc22e2d2f2a2 Whats New In Critical Illness And Injury Science The Quest For Effective And Safe Co Induction Agents In Spontaneously Breathing Patients Undergoing General Anesthesia Pdf
Migraine Disordersdrug therapy Substances.
8496065 PubMed - indexed for MEDLINE Publication Types. Migraine Disordersdrug therapy Substances. Sleep problems insomnia. Nasal spray solution 1mg If you are experiencing a medical crisis please call 911 or contact your local emergency assistance service immediately. It is used to treat moderate to severe pain. Prior Approval is required only in order to EXCEED the standard allowance Stadol Nasal Spray butorphanol NOTE. It is also used to prevent pain before surgery and during child birth. Butorphanoladministration. This is not a complete list of side effects and others may occur.
09 Sodium Chloride Small Volume Bags 150 mL Apr 15 2021. You may report side effects to FDA at 1-800-FDA-1088. Sleep problems insomnia. BUTORPHANOL is a pain reliever. The efficacy of BUTORPHANOL Nasal Spray for periods longer than 3 days has not been established. If the patient requires more than the quantity above per 90 days a prior approval is required. Nasal spray solution 1mg If you are experiencing a medical crisis please call 911 or contact your local emergency assistance service immediately.



























Post a Comment for "Stadol Nasal Spray Discontinued"